Design of soft material dynamics
The ability to impart precisely controlled dynamic processes into soft materials is a grand challenge in soft materials design. Polymers comprise dynamics ranging from local monomer motions to chain diffusion to bulk flow, all of which can be impacted by a myriad of factors including the polymer chemistry, architecture, molecular weight, dynamic interactions (ions, H-bonds, reversible covalent bonds), and the glass or melting temperatures. These dynamics are relevant for molecular transport (electrolytes, membranes, drug delivery), interactions with substrates (pressure sensitive adhesives), additive manufacturing, tissue engineering, and energy dissipation.
A current focus in the group is how to rationally incorporate dynamic bonds into polymer networks to precisely control where relaxation processes occur. By controlling the architecture of the networks, we have discovered that two dynamic bonds will only give one relaxation mode if they in a “telechelic” network were only two points connect each strand to the rest of the system. Conversely, “pendant” networks allow us to impart multiple relaxation modes from multiple dynamic bonds. The faster process is well defined and occurs in the same location regardless of the slower bonds. We are also working to understand how mixed bond exchange mechanisms allow for orthogonal design of dynamic processes in polymer networks.

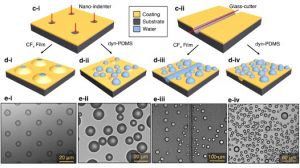
The viscoelastic properties of dynamics networks are also key to their thin film properties including adhesion, wetting, and self-healing. We have developed ultrathin silicone networks which show self-healing in response to a scratch or pinhole defect much faster than in the bulk. They also can exhibit superhydrophobic behavior and long term durability due to the presence of dynamic bonds. We are working to understand the fundamental mechanisms of how bond exchange impacts thin film physics.